
They also built waste repositories, dams, water filtration systems and the “sarcophagus”, which entombs the entire fourth reactor to contain the remaining radioactive material.ġ4. They worked on decontamination and major construction projects, including the establishment of settlements and towns for plant workers and evacuees. These 600,000 individuals received special benefits because of their involvement, on- and off-site, in tackling the accident’s aftermath. The Russian registries list approximately 400,000 liquidators as of 1991 and approximately 600,000 people were granted the status of “liquidator”. The exact number of liquidators is unknown because there are no completely accurate records of the people involved in the clean-up. These workers were mostly plant employees, Ukrainian fire-fighters plus many soldiers and miners from Russia, Belarus, Ukraine and other parts of the former Soviet Union. How was this area cleaned up after the accident?Įmergency workers (liquidators) were drafted into the area and helped to clean up the plant premises and the surrounding area. The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. The cookie is used to store the user consent for the cookies in the category "Performance". 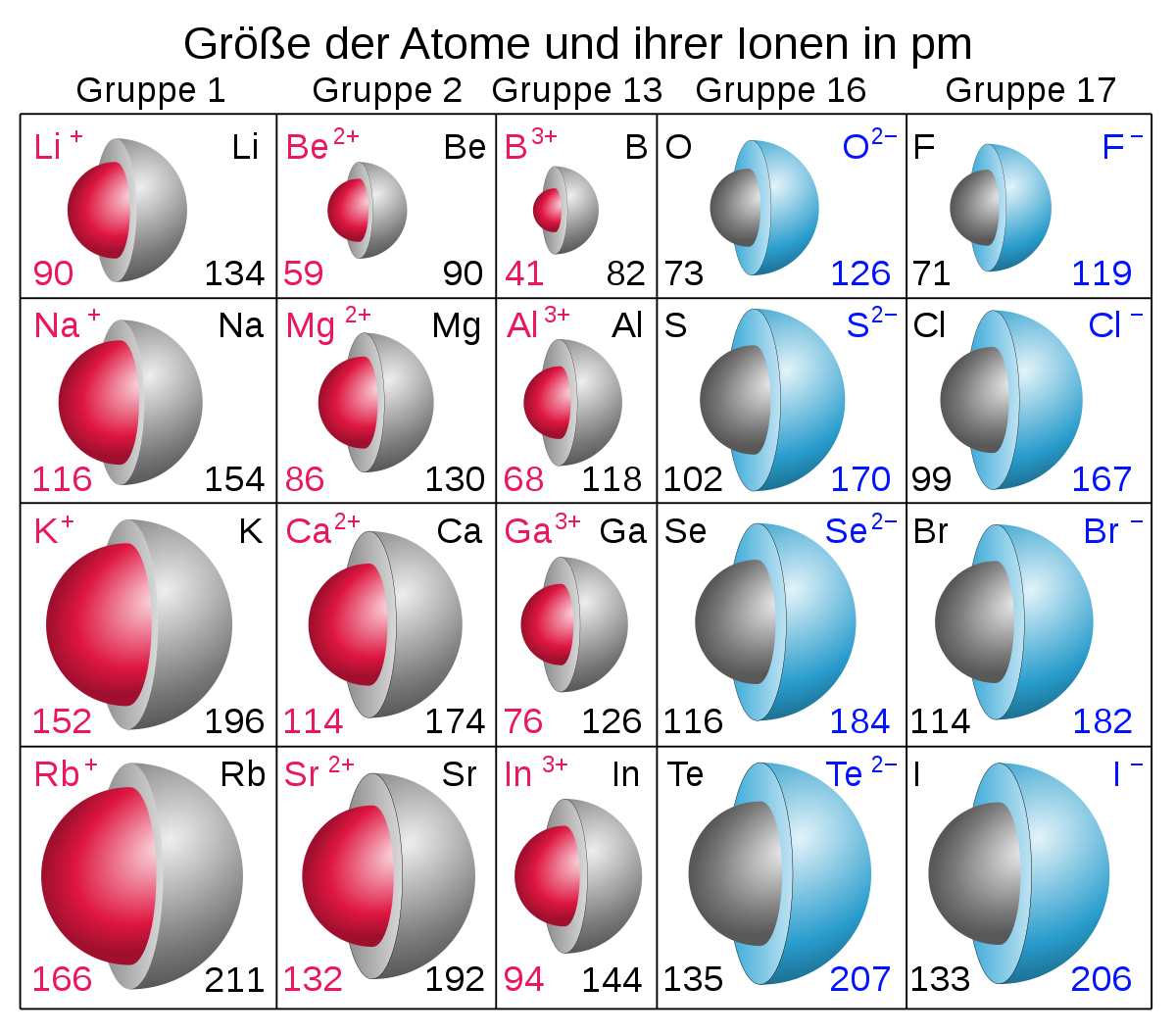 
This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. The cookies is used to store the user consent for the cookies in the category "Necessary". The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. Helium is a chemical element with atomic number 2 which means there are 2 protons and 2 electrons in the atomic structure. Its monatomic form (H) is the most abundant chemical substance in the Universe, constituting roughly 75% of all baryonic mass. Which is the most abundant chemical element in the universe? What’s the atomic radius of phosphorus and silicon?Ītomic radius of Silicon (Si) 210 pm. What is the atomic radius of boron and lithium?Ītomic radius of Lithium (Li) 182 pm. Thus, helium is the smallest element, and francium is the largest. Which is the smallest element with the largest atomic radius?Īs can be seen in the figures below, the atomic radius increases from top to bottom in a group, and decreases from left to right across a period. Thus electrons in oxygen atom will be closer to nucleus than in the case of nitrogen. Also, oxygen has same number of shells as nitrogen. The eight protons in oxygen atom’s nucleus will exert more force than nitrogen’s seven electrons. Oxygen has eight electrons in its shells. Why is the atomic radius of oxygen larger than nitrogen? That means its electrons are more strongly attracted to its nucleus, hence the electronic cloud is more compressed, which results in a smaller size. Why does carbon have a larger atomic radius than oxygen?īoth are in the same period, but oxygen has a higher nuclear effective charge than carbon. Helium/Van der Waals radius What is the increasing atomic radius of carbon? As we can see, only Potassium has four electron shells, therefore Potassium has the largest atomic size. 
The potassium atom has an extra electron shell compared to the sodium atom, which means its valence electrons are further from the nucleus, giving potassium a larger atomic radius.ĭoes potassium have a larger atomic radius than oxygen?Īn atom’s size is defined by the distance from the nucleus to the valence (outer) shell of electrons. Does potassium have a larger atomic radius? Since C comes before O in the second period, O will have a smaller atomic radius than C. We know that atomic radius decreases from left to right within a period due to the increase in effective nuclear charge. Which has a greater atomic radius carbon or oxygen? As can be seen in the figures below, the atomic radius increases from top to bottom in a group, and decreases from left to right across a period. Which element has the largest atomic radius list?Ītomic radii vary in a predictable way across the periodic table. 8 Which is the most abundant chemical element in the universe?.7 What is the atomic radius of boron and lithium?.6 What is the atomic radius of nitrogen?.4 What is the increasing atomic radius of carbon?.3 What is the atomic radius of potassium?.2 Does potassium have a larger atomic radius?.1 Which element has the largest atomic radius list?.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
